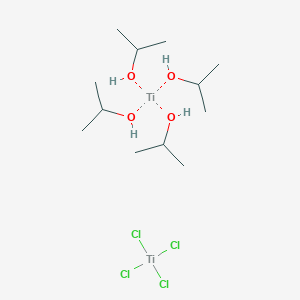
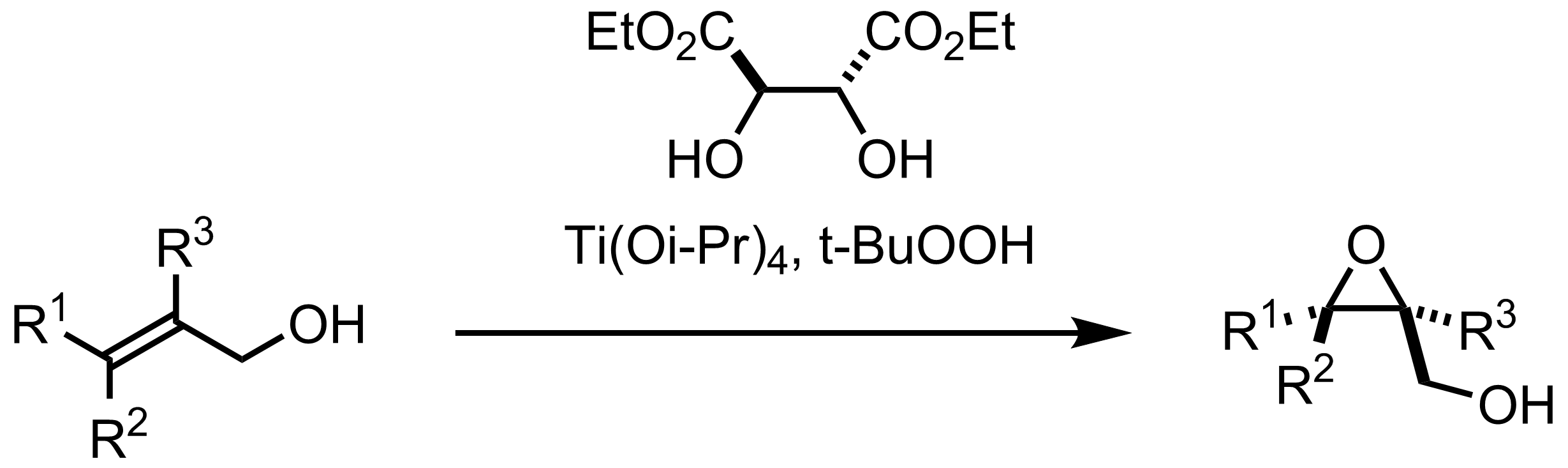
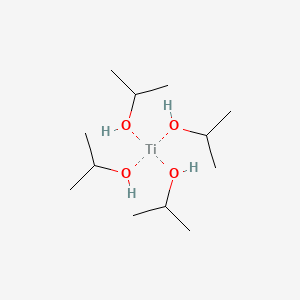
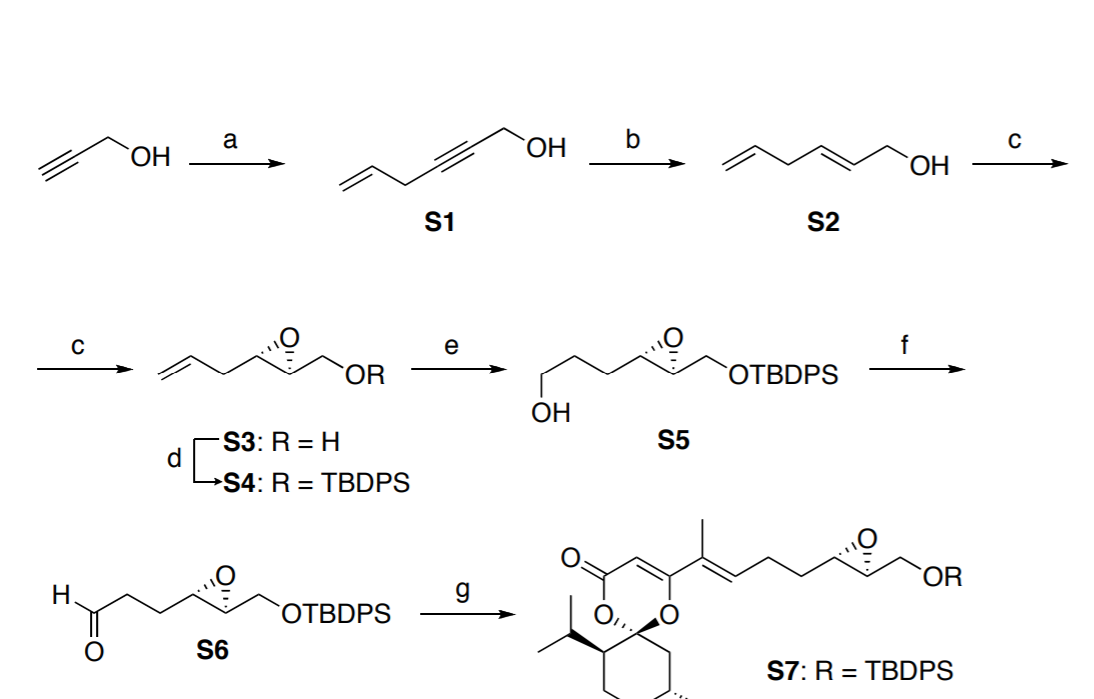
Reagents and conditions (i) Diethyl–L-tartrate, Ti(OiPr)4, t-BuOOH,... | Download Scientific Diagram
![Can the Ti(OiPr) 4 / n BuLi combination of reagents function as a catalyst for [2+2+2] alkyne cyclotrimerisation reactions? - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C8NJ04931A Can the Ti(OiPr) 4 / n BuLi combination of reagents function as a catalyst for [2+2+2] alkyne cyclotrimerisation reactions? - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C8NJ04931A](https://pubs.rsc.org/image/article/2018/NJ/c8nj04931a/c8nj04931a-s1_hi-res.gif)
Can the Ti(OiPr) 4 / n BuLi combination of reagents function as a catalyst for [2+2+2] alkyne cyclotrimerisation reactions? - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C8NJ04931A

Ti(Oi-Pr)4-promoted photoenolization Diels–Alder reaction to construct polycyclic rings and its synthetic applications | Nature Communications
![Ti(OiPr) 4 / n BuLi: an attractive reagent system for [2+2+2] cyclotrimerisation reactions - Chemical Communications (RSC Publishing) DOI:10.1039/C4CC02698E Ti(OiPr) 4 / n BuLi: an attractive reagent system for [2+2+2] cyclotrimerisation reactions - Chemical Communications (RSC Publishing) DOI:10.1039/C4CC02698E](https://pubs.rsc.org/image/article/2014/CC/c4cc02698e/c4cc02698e-s1_hi-res.gif)
Ti(OiPr) 4 / n BuLi: an attractive reagent system for [2+2+2] cyclotrimerisation reactions - Chemical Communications (RSC Publishing) DOI:10.1039/C4CC02698E
![Can the Ti(OiPr) 4 / n BuLi combination of reagents function as a catalyst for [2+2+2] alkyne cyclotrimerisation reactions? - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C8NJ04931A Can the Ti(OiPr) 4 / n BuLi combination of reagents function as a catalyst for [2+2+2] alkyne cyclotrimerisation reactions? - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C8NJ04931A](https://pubs.rsc.org/image/article/2018/NJ/c8nj04931a/c8nj04931a-s2_hi-res.gif)
Can the Ti(OiPr) 4 / n BuLi combination of reagents function as a catalyst for [2+2+2] alkyne cyclotrimerisation reactions? - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C8NJ04931A

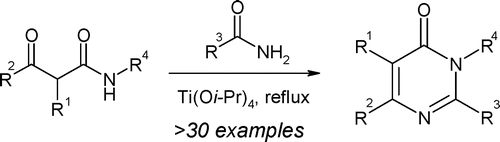
Multi-step Split Synthesis on the TridentTM: Parallel Solution-Phase Synthesis of Urea and Amide Library

Hydrosiloxane–Ti(OiPr)4: an efficient system for the reduction of primary amides into primary amines as their hydrochloride salts - ScienceDirect

Decomposition Pathways of Titanium Isopropoxide Ti(OiPr)4: New Insights from UV-Photodissociation Experiments and Quantum Chemical Calculations | The Journal of Physical Chemistry A

Novel Knöevenagel-type reaction via titanium enolate derived from Ti(O-i-Pr) 4 and diketene - ScienceDirect