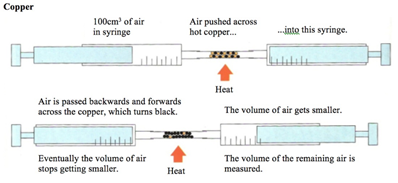
Oxygen and oxides 2.16 recall the gases present in air and their approximate percentage by volume - ppt video online download
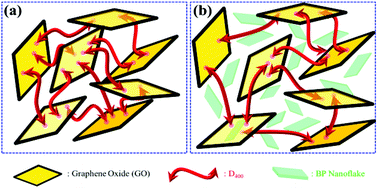
Graphene oxide/black phosphorus nanoflake aerogels with robust thermo-stability and significantly enhanced photothermal properties in air - Nanoscale (RSC Publishing)

Enhancing thermal oxidation and fire resistance of reduced graphene oxide by phosphorus and nitrogen co-doping: Mechanism and kinetic analysis - ScienceDirect
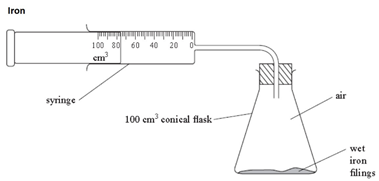
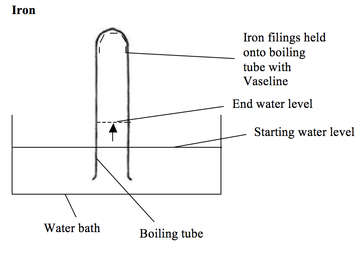
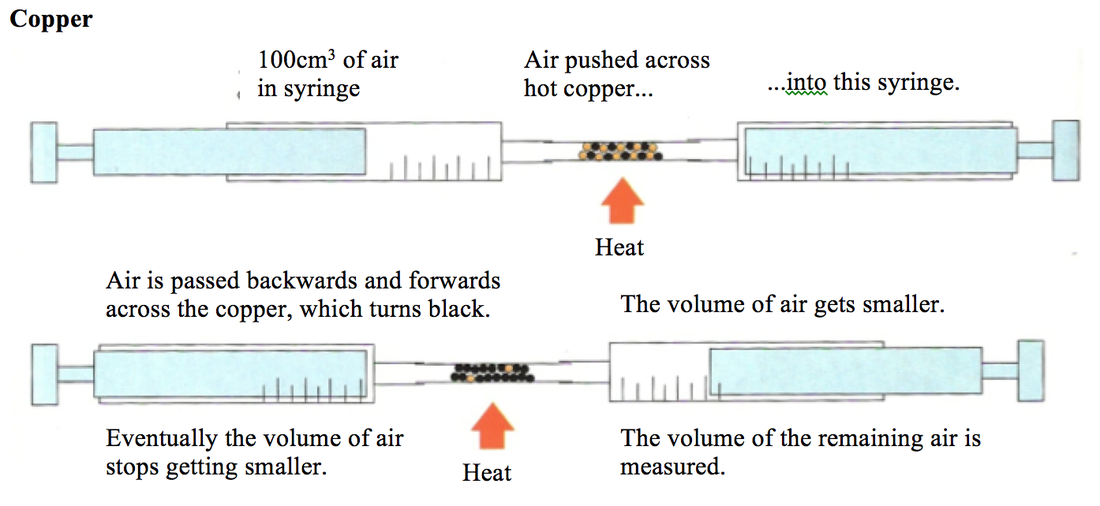
2:10 understand how to determine the percentage by volume of oxygen in air using experiments involving the reactions of metals (e.g. iron) and non-metals (e.g. phosphorus) with air - TutorMyself Chemistry
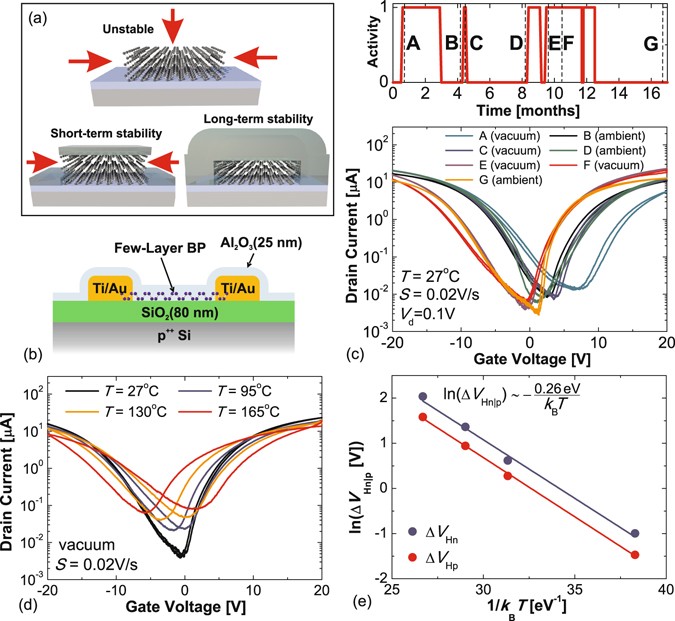
Highly-stable black phosphorus field-effect transistors with low density of oxide traps | npj 2D Materials and Applications

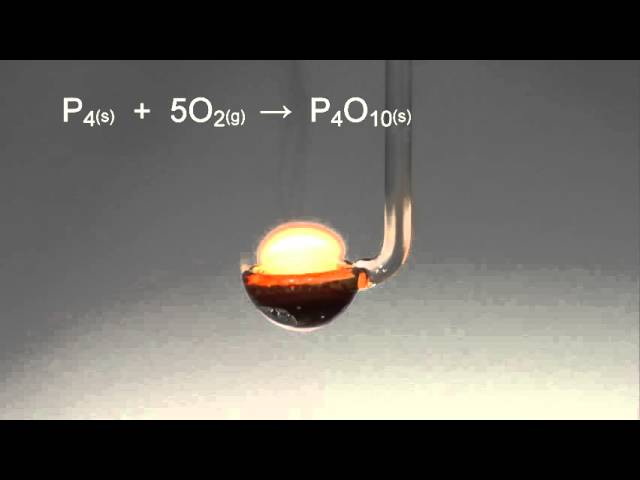
Translate the following statement into chemical equations and then balance the equations: (i) Phosphorus burns in oxygen to give phosphorus pentoxide. (ii) Aluminium metal replaces iron from ferric oxide, Fe(2)O(3), giving aluminium
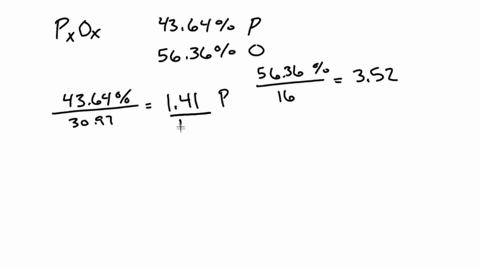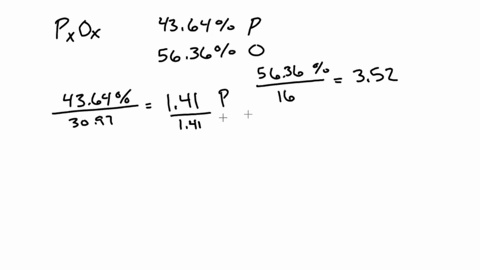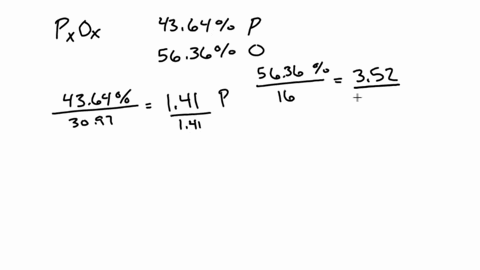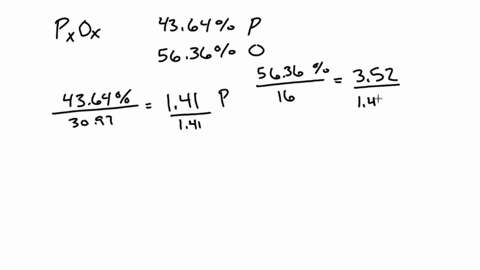
SOLVED:A 1.45-g sample of phosphorus burns in air and forms 2.57 g of a phosphorus oxide. Calculate the empirical formula of the oxide.
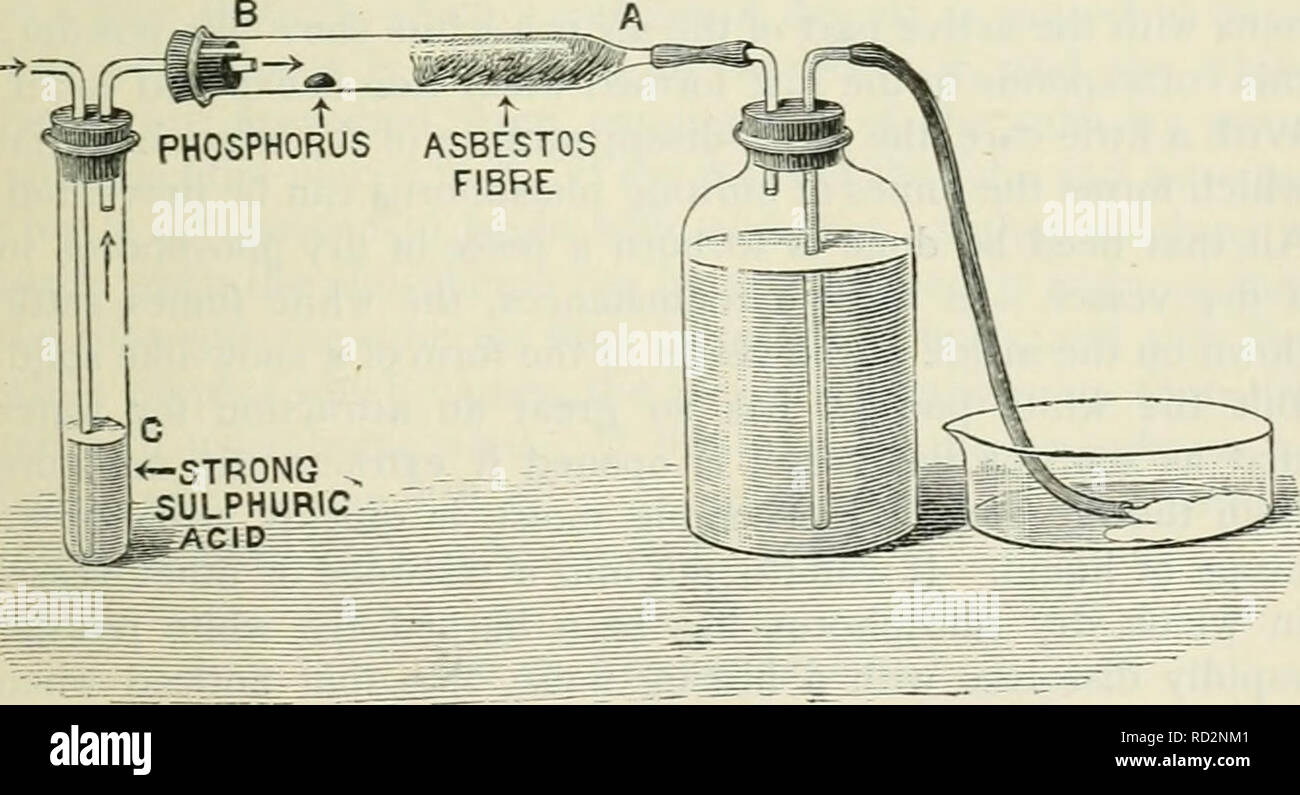
Elementary physics and chemistry: second stage. Science. BURNING PHOSPHORUS IN AIR. 109 powder is deposited upon the sides of the cylinder. When the phosphorus has ceased to burn, hft up the