Postoperative fondaparinux versus preoperative enoxaparin for prevention of venous thromboembolism in elective hip-replacement surgery: a randomised double-blind comparison - The Lancet

Natural killer cells and immune-checkpoint inhibitor therapy: Current knowledge and new challenges: Molecular Therapy - Oncolytics

Internal mammary and medial supraclavicular lymph node chain irradiation in stage I–III breast cancer (EORTC 22922/10925): 15-year results of a randomised, phase 3 trial - The Lancet Oncology
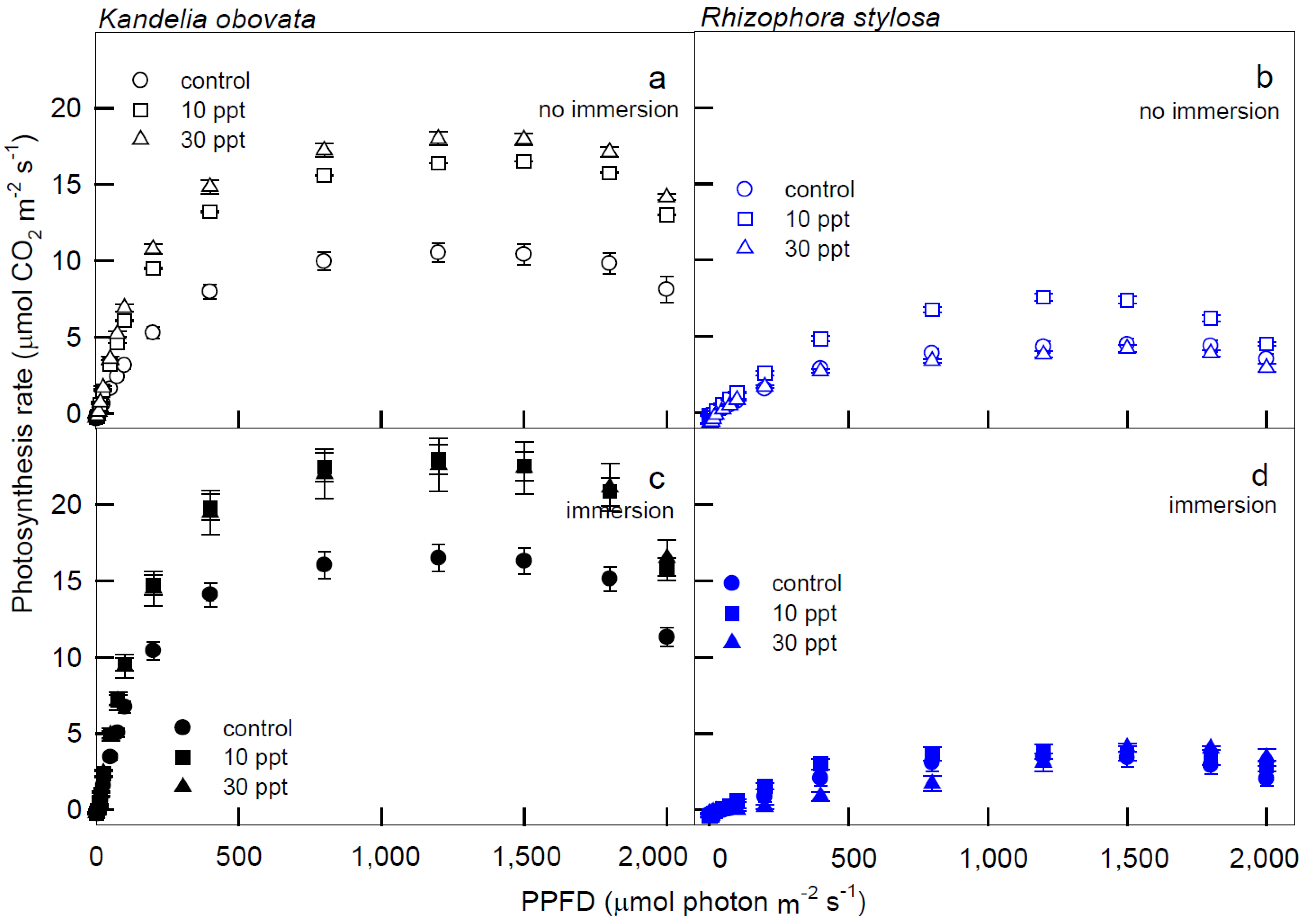
Cells | Free Full-Text | Photosynthesis in Response to Different Salinities and Immersions of Two Native Rhizophoraceae Mangroves

Rucaparib versus standard-of-care chemotherapy in patients with relapsed ovarian cancer and a deleterious BRCA1 or BRCA2 mutation (ARIEL4): an international, open-label, randomised, phase 3 trial - The Lancet Oncology

The clinical and cost-effectiveness of total versus partial knee replacement in patients with medial compartment osteoarthritis (TOPKAT): 5-year outcomes of a randomised controlled trial - The Lancet

Addition of cetuximab to oxaliplatin-based first-line combination chemotherapy for treatment of advanced colorectal cancer: results of the randomised phase 3 MRC COIN trial - The Lancet

Solid-State NMR Investigations of Extracellular Matrixes and Cell Walls of Algae, Bacteria, Fungi, and Plants | Chemical Reviews

Amazon.com: Mini VCI J2534 Tis Techstream Cable OBD2 Diagnostic Cable for Supporting TIS Techstream : Automotive

Rituximab versus intravenous cyclophosphamide in patients with connective tissue disease-associated interstitial lung disease in the UK (RECITAL): a double-blind, double-dummy, randomised, controlled, phase 2b trial - The Lancet Respiratory Medicine

Antibody-mediated allograft rejection is associated with an increase in peripheral differentiated CD28-CD8+ T cells – Analyses of a cohort of 1032 kidney transplant recipients - eBioMedicine

LncRNA Osilr9 coordinates promoter DNA demethylation and the intrachromosomal loop structure required for maintaining stem cell pluripotency: Molecular Therapy

Enforced expression of Runx3 improved CAR-T cell potency in solid tumor via enhancing resistance to activation-induced cell death: Molecular Therapy

Patient-reported outcomes with durvalumab after chemoradiotherapy in stage III, unresectable non-small-cell lung cancer (PACIFIC): a randomised, controlled, phase 3 study - The Lancet Oncology

Tirzepatide versus insulin glargine in type 2 diabetes and increased cardiovascular risk (SURPASS-4): a randomised, open-label, parallel-group, multicentre, phase 3 trial - The Lancet

Exons 45–55 Skipping Using Mutation-Tailored Cocktails of Antisense Morpholinos in the DMD Gene: Molecular Therapy

Temozolomide chemotherapy versus radiotherapy in high-risk low-grade glioma (EORTC 22033-26033): a randomised, open-label, phase 3 intergroup study - The Lancet Oncology

Lisocabtagene maraleucel versus standard of care with salvage chemotherapy followed by autologous stem cell transplantation as second-line treatment in patients with relapsed or refractory large B-cell lymphoma (TRANSFORM): results from an interim
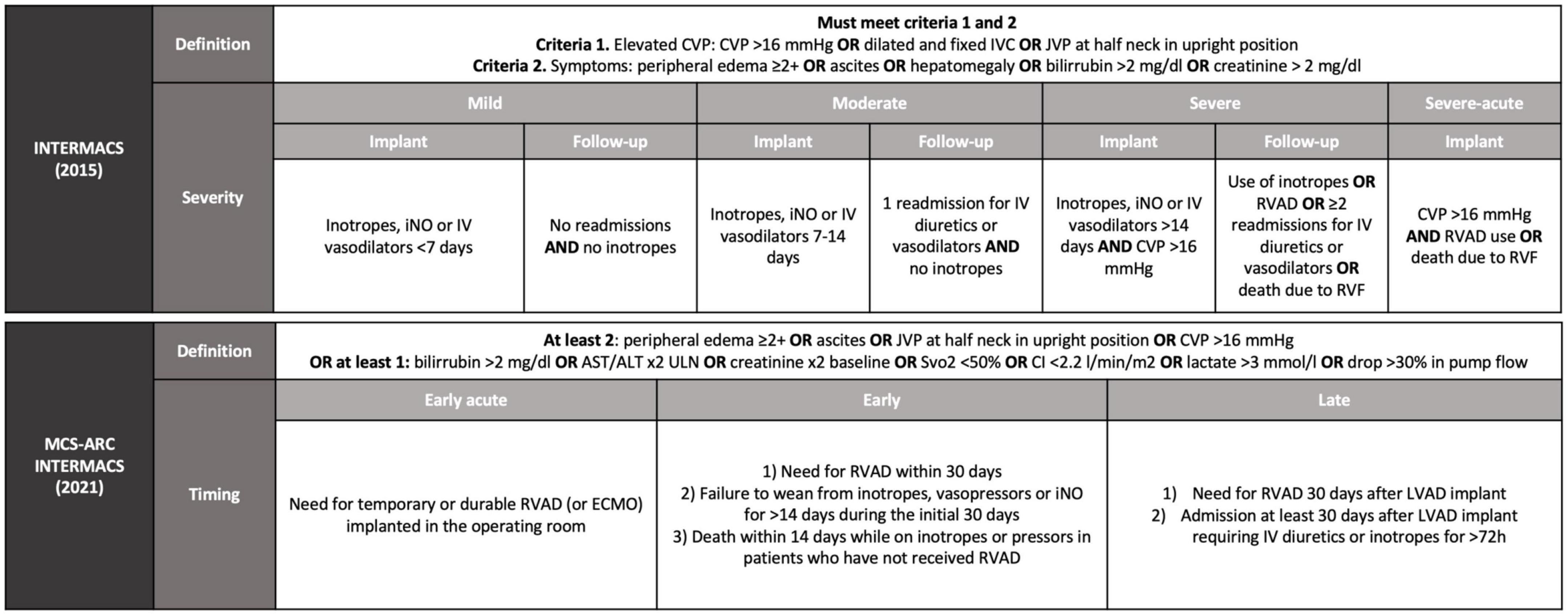
Frontiers | Prediction, prevention, and management of right ventricular failure after left ventricular assist device implantation: A comprehensive review

Late Effects of Severe Acute Graft-versus-Host Disease on Quality of Life, Medical Comorbidities, and Survival - Transplantation and Cellular Therapy, Official Publication of the American Society for Transplantation and Cellular Therapy

Chemical Transformations of Biomass-Derived C6-Furanic Platform Chemicals for Sustainable Energy Research, Materials Science, and Synthetic Building Blocks | ACS Sustainable Chemistry & Engineering
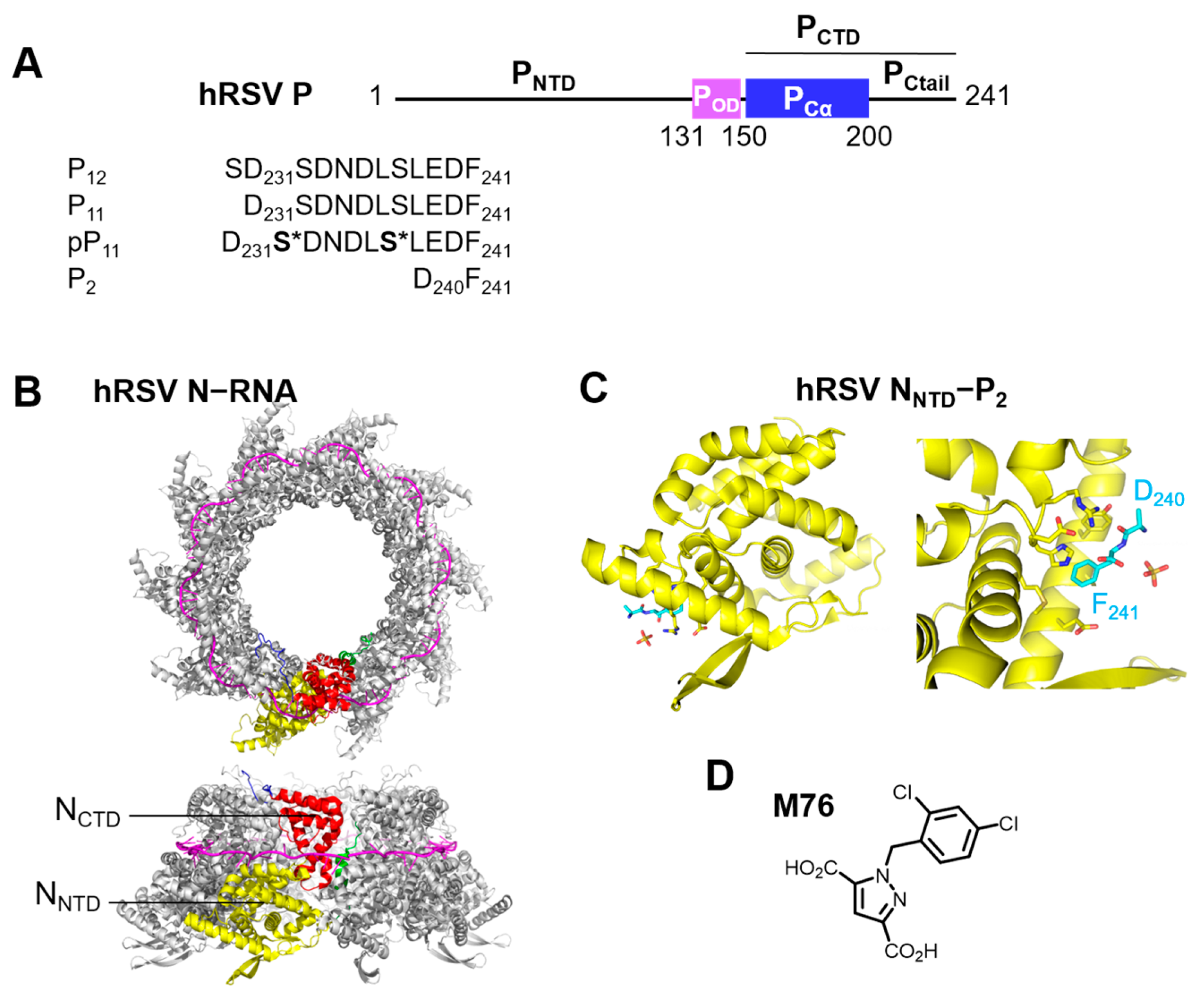
IJMS | Free Full-Text | Investigation of the Fuzzy Complex between RSV Nucleoprotein and Phosphoprotein to Optimize an Inhibition Assay by Fluorescence Polarization

Seven days treatment with the angiotensin II type 2 receptor agonist C21 in hospitalized COVID-19 patients; a placebo-controlled randomised multi-centre double-blind phase 2 trial - eClinicalMedicine

National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: IV. The 2020 Highly morbid forms report - Transplantation and Cellular Therapy, Official Publication of the