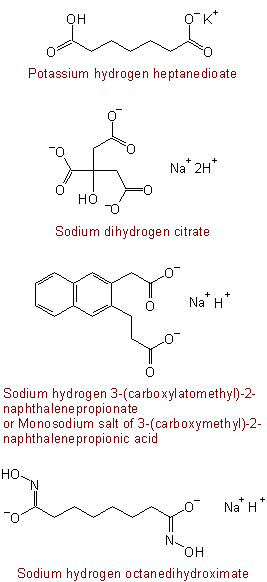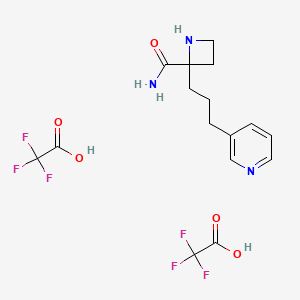
Buy 2-(3-Pyridin-3-yl-propyl)-azetidine-2-carboxylic acid amidedi(trifluoroacetic acid salt) - 1361111-96-7 | Benchchem
![1R,3S,5R)-2-Azabicyclo[3.3.0]octane-3-carboxylic Acid, Benzyl Ester p-Toluenesulphonic Acid Salt | SCBT - Santa Cruz Biotechnology 1R,3S,5R)-2-Azabicyclo[3.3.0]octane-3-carboxylic Acid, Benzyl Ester p-Toluenesulphonic Acid Salt | SCBT - Santa Cruz Biotechnology](https://media.scbt.com/product/1r-3s-5r-2-azabicyclo3-3-0octane-3-carboxylic-acid-benzyl-ester-p-toluenesulphonic-acid-salt-_08_27_b_82768.jpg)
1R,3S,5R)-2-Azabicyclo[3.3.0]octane-3-carboxylic Acid, Benzyl Ester p-Toluenesulphonic Acid Salt | SCBT - Santa Cruz Biotechnology

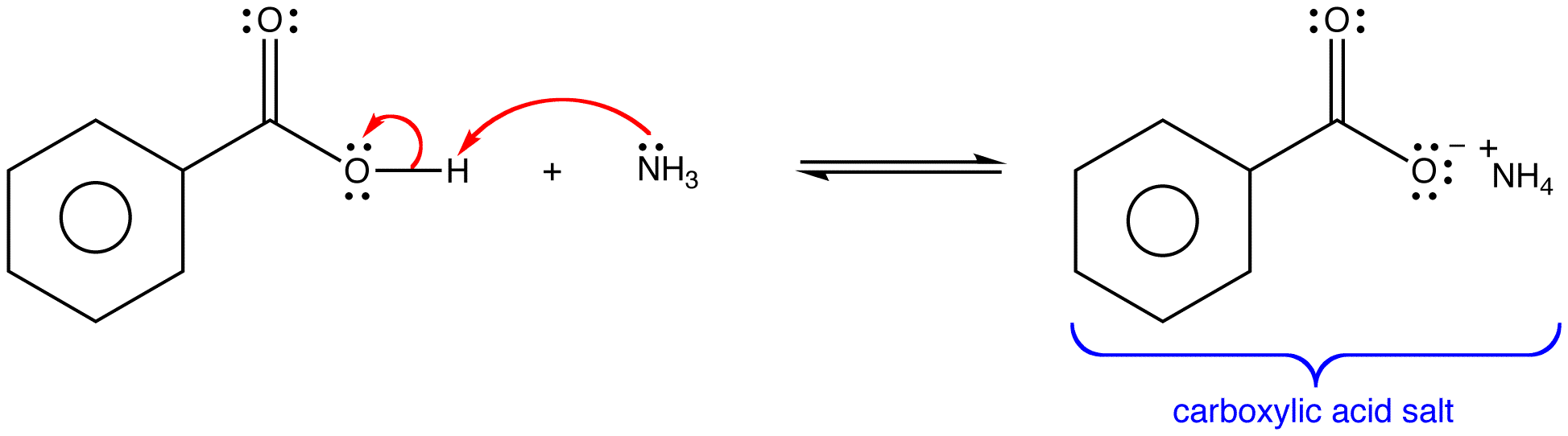
Write a chemical equation for the conversion of the following carboxylic acid salt to its parent carboxylic acid. Let hydrochloric acid (HCl) be the source of the needed hydronium ions. Sodium lactate.

Write a chemical equation for the conversion of the following carboxylic acid salt to its parent carboxylic acid. Let hydrochloric acid (HCl) be the source of the needed hydronium ions. Sodium lactate.
![1R,4R,5R)-3-Oxo-2-oxabicyclo[2.2.1]heptane-5-carboxylic acid cinchonidine salt 97% | CAS: 1233219-43-6 | AChemBlock 1R,4R,5R)-3-Oxo-2-oxabicyclo[2.2.1]heptane-5-carboxylic acid cinchonidine salt 97% | CAS: 1233219-43-6 | AChemBlock](http://inter.achemblock.com/media/catalog/L21307.png)
1R,4R,5R)-3-Oxo-2-oxabicyclo[2.2.1]heptane-5-carboxylic acid cinchonidine salt 97% | CAS: 1233219-43-6 | AChemBlock

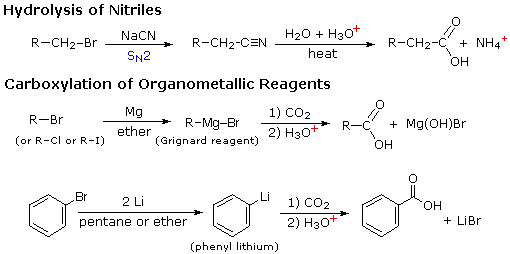

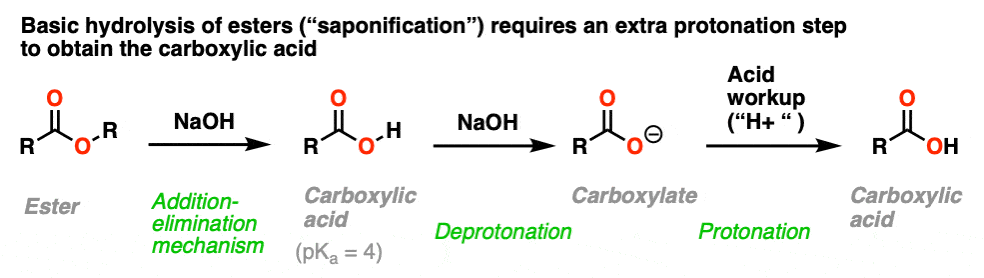

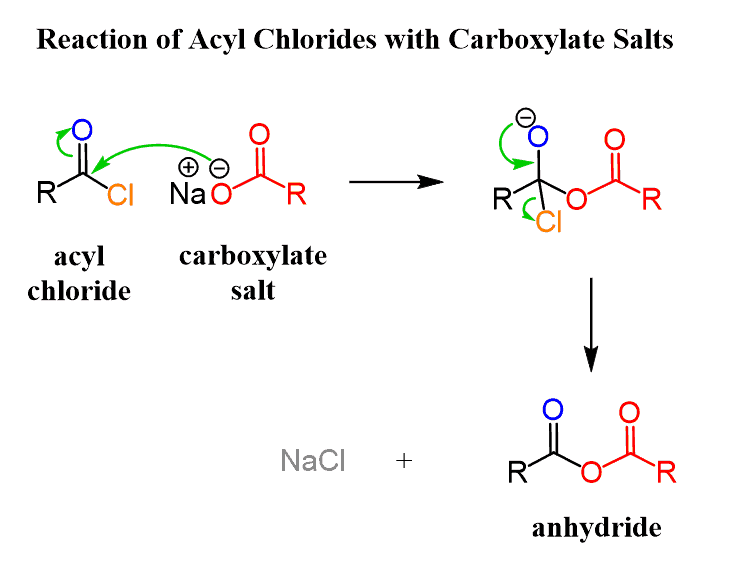
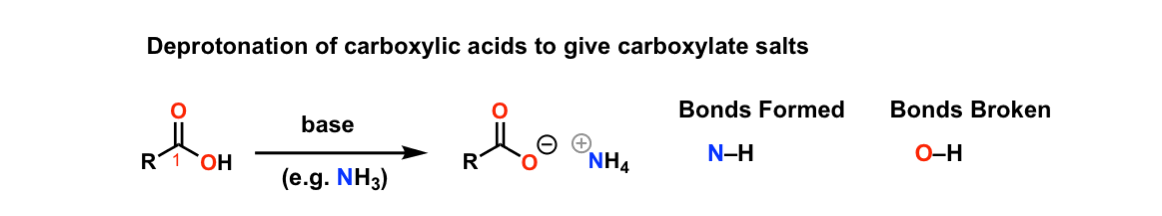

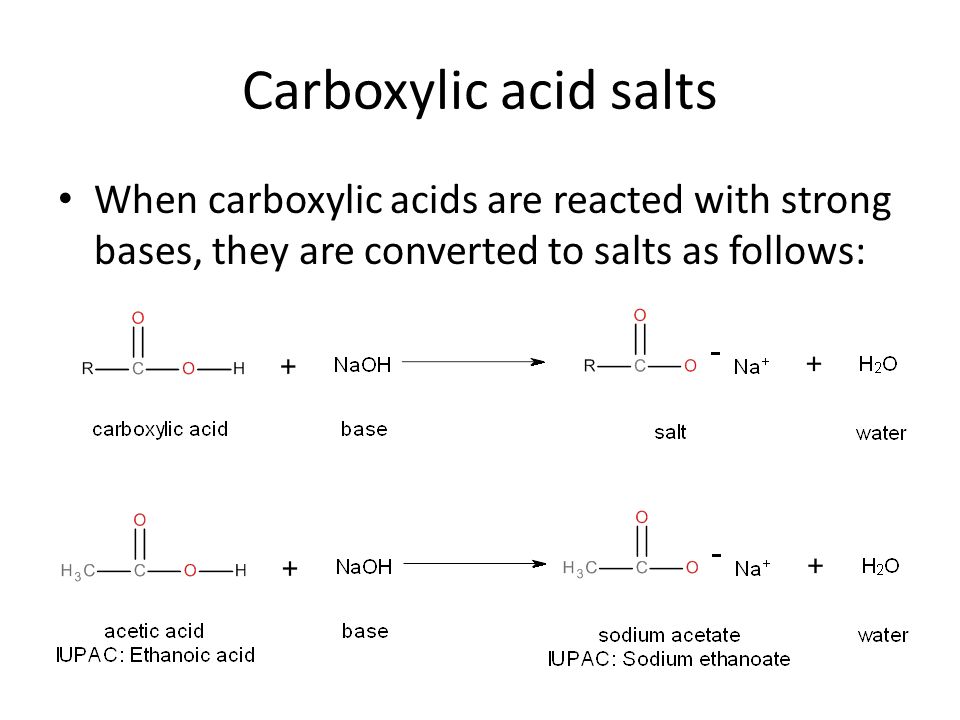

![2-Azaspiro[3.3]heptane-6-carboxylic acid TFA salt 97% | CAS: 1523572-02-2 | AChemBlock 2-Azaspiro[3.3]heptane-6-carboxylic acid TFA salt 97% | CAS: 1523572-02-2 | AChemBlock](http://inter.achemblock.com/media/catalog/C-3540.png)